BeNa Culture Collection
-
Login |
-
EngLish

 info@bncc.com
info@bncc.com
BeNa Culture Collection

 info@bncc.com
info@bncc.com



| Culture medium | BNCC352251 |
| Description | YM agar medium |
| Composition | Yeast Extract Powder:3.0g, Malt Extract:3.0g, Glucose:10.0g, Casein Peptone:5.0g, Agar:20.0g, pH:6.2±0.2(25℃) |
| Growth conditions | 28°C; 24-48h; Aerobic; |
| Storage conditions | -20°C |
| morphology | Milky white, convex, moist, viscous, Gram-positive cocci |
| Sharing mode | Public welfare sharing |
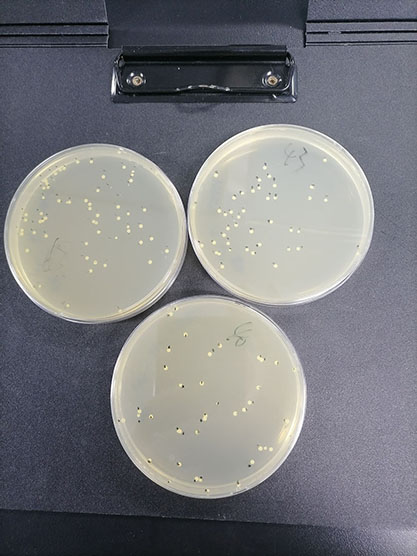
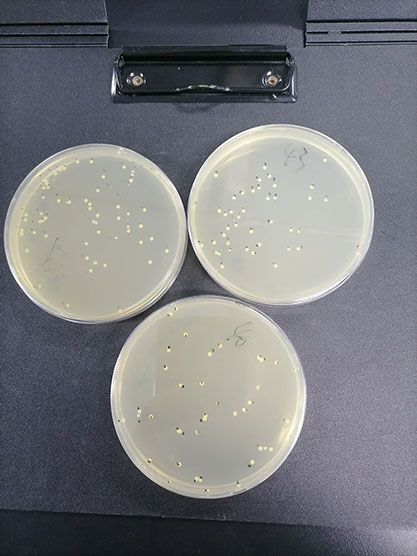
 Quantitative strains of Bacillus subtilis subsp(Quantitative strains of Bacillus subtilis subsp)
BNCC367736
Quantitative strains of Bacillus subtilis subsp(Quantitative strains of Bacillus subtilis subsp)
BNCC367736
 Influenza A virus(H3N2) nucleic acid reference (Heat inactivated) (Strongly positive)(Influenza A virus(H3N2) nucleic acid reference (Heat inactivated) (Strongly positive))
BNCC381677
Influenza A virus(H3N2) nucleic acid reference (Heat inactivated) (Strongly positive)(Influenza A virus(H3N2) nucleic acid reference (Heat inactivated) (Strongly positive))
BNCC381677
 Japanese encephalitis virus antigen test reference (Chemical inactivation) (Strongly positive)(Japanese encephalitis virus antigen test reference (Chemical inactivation) (Strongly positive))
BNCC394528
Japanese encephalitis virus antigen test reference (Chemical inactivation) (Strongly positive)(Japanese encephalitis virus antigen test reference (Chemical inactivation) (Strongly positive))
BNCC394528
 Human herpesvirus 3 DNA lab quality control(varicella-zoster virus)(Human herpesvirus 3 DNA lab quality control(varicella-zoster virus))
BNCC395778
Human herpesvirus 3 DNA lab quality control(varicella-zoster virus)(Human herpesvirus 3 DNA lab quality control(varicella-zoster virus))
BNCC395778
 JC polyomavirus DNA lab quality control(JC polyomavirus DNA lab quality control)
BNCC394335
JC polyomavirus DNA lab quality control(JC polyomavirus DNA lab quality control)
BNCC394335
 Canine parainfluenza virus nucleic acid reference (Heat inactivated) (Strongly positive)(Canine parainfluenza virus nucleic acid reference (Heat inactivated) (Strongly positive))
BNCC393916
Canine parainfluenza virus nucleic acid reference (Heat inactivated) (Strongly positive)(Canine parainfluenza virus nucleic acid reference (Heat inactivated) (Strongly positive))
BNCC393916